Stefan Andersson Engels is an SFI Research Fellow, Senior Staff Researcher and PI at Tyndall specialising in Biophotonics Research.
Throughout his fruitful career, Stefan has accumulated deep expertise that addresses major biomedical global challenges. This, he has demonstrated through significant deep-tech innovation and impact in biomedical science through ground-breaking biophotonics research. Along his journey, his dedication to teaching has helped to secure and equip the next generation of biophotonics research leaders. Stefan shares his path to Research Excellence in Biophotonics research.
As a kid I had always known I would become a researcher. I was so impressed by their expertise, which I discovered mostly from Radio and TV interviews. When entering Lund University,Sweden, I had to make a choice between Physics and Medicine. In the end I selected Physics as it was the most challenging education program, and it was better suited to my learning skills. I was offered a PhD position in two programs and chose Laser spectroscopy for medical applications. Lund University was pioneering research in the field Biomedical Optics and it was an incredible experience to be part of the birth of a new field. Within seven years after my PhD I was a fully tenured Professor at Lund and in 2016 accepted a position at IPIC, Tyndall National Institute at UCC. My focus now is on building and mentoring a dynamic new biophotonics team who are working on clinical translation research, fundamental research for deep tissue imaging based on acousto-optics or upconverting nanoparticles and wearable sensors.
I am privileged to be and have been engaged in the most stimulating biophotonics research and mentorship activities, including:
My entire career has been dedicated to developing fluorescence-based techniques and technologies for medical diagnostics and surgical guidance using both point-spectroscopy and multispectral imaging techniques.
This specialised work is and has been conducted in close collaboration with clinicians in preclinical and clinical studies. The work resulted also in a key patent and spin-out that became known as SpectraPhos which I co-founded. The company and IP has been acquired in several steps, first by Xillix Inc. in Canada, then by Novadaq and later Stryker. My PhD thesis entitled ‘Laser-Induced Fluorescence for Medical Diagnostics’ was published in 1989 and is based on 16 publications presenting preclinical and clinical studies on laser-induced fluorescence and photodynamic therapy of malignant tumours and atherosclerotic plaques.
Relevant research examples and selected publications include:
.jpg)
S. Andersson-Engels, J. Johansson, K. Svanberg, S. Svanberg, Fluorescence imaging and point measurements of tissue applications to the demarcation of malignant tumors and atherosclerotic plaques from normal tissue, Photochemistry and Photobiology, 53, 807-814 (1991). Cited by 246 This is a very early review of the field of laser-induced fluorescence for medical diagnostics application. It describes the research conducted to use fluorescence as a method to help visualise malignant tumours and disease-modified blood vessels in the management of oncological and vascular diseases.
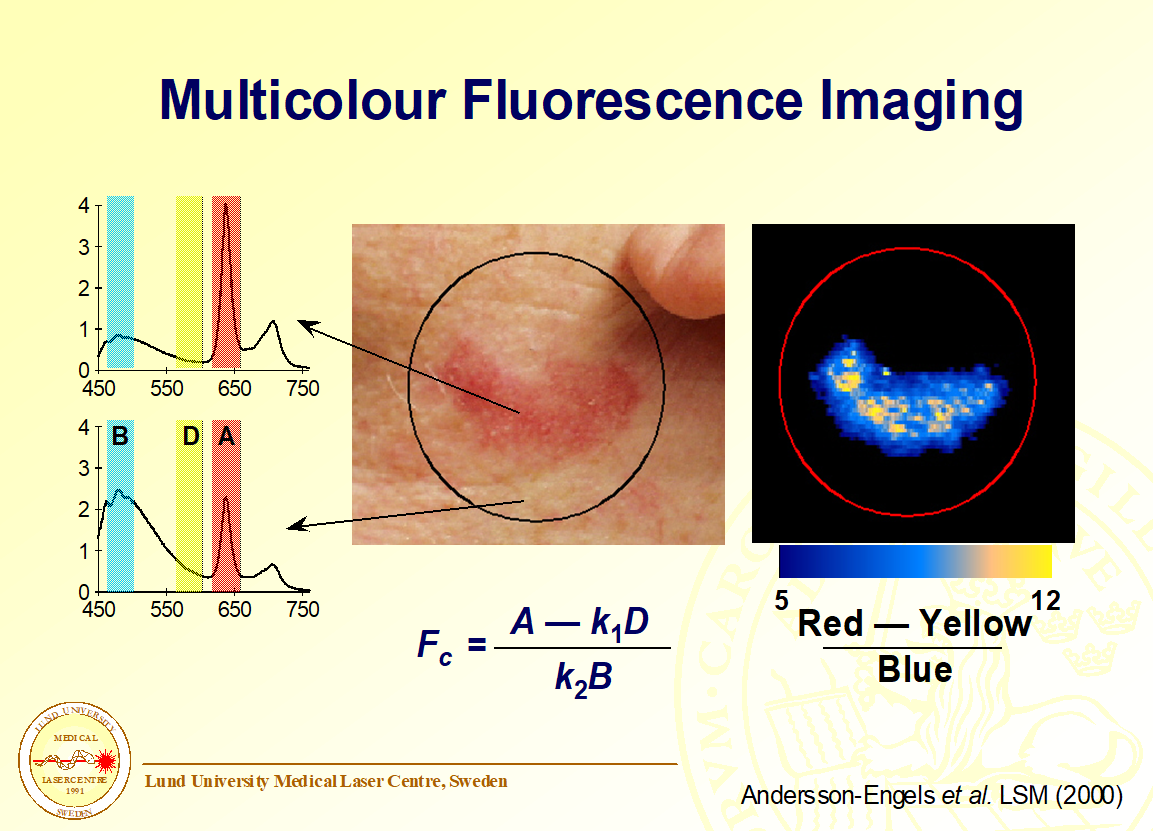
S S. Andersson-Engels, J. Johansson, S. Svanberg, Medical Diagnostic System Based On Simultaneous Multispectral Fluorescence Imaging, Applied Optics, 33 (1994) 8022-8029. Cited by 109 Here we presented a multispectral fluorescence imaging system developed for medical diagnostics. This is the system that SpectraPhos based their commercial product on. The publication describes the system as well as its performance characterization.
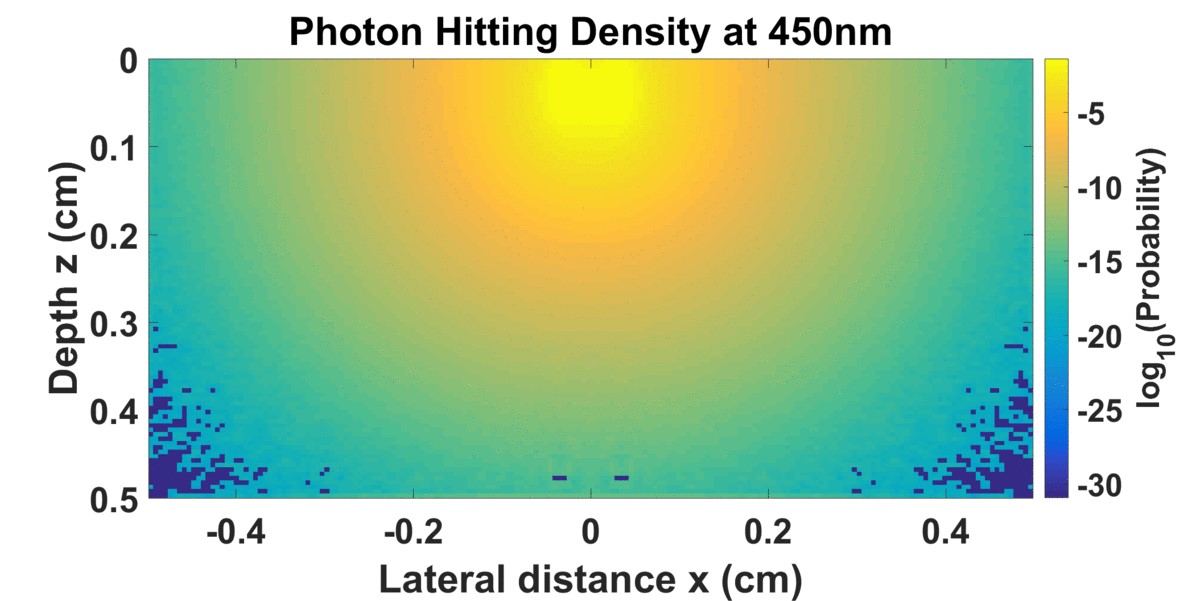
S. Andersson-Engels, C. af Klinteberg, K. Svanberg, S. Svanberg, In vivo fluorescence imaging for tissue diagnostics, Physics in Medicine and Biology, 42 (1997) 815-824. Cited by 329 In this review of the biomedical fluorescence diagnostic field, we carefully discuss how the measured fluorescence signal is influenced by the intrinsic fluorescence properties as well as of how the measured signals are influenced by the attenuation of both the excitation and emission light.
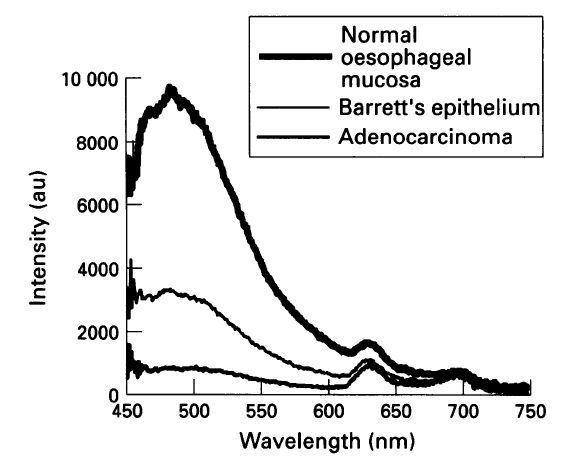
C.S. Von Holstein, A.M.K. Nilsson, S. Andersson-Engels, R. Willen, B. Walther, K. Svanberg, Detection of adenocarcinoma in Barrett's oesophagus by means of laser induced fluorescence, Gut, 39 (1996) 711-716. Cited by 101 This clinical study provides rather comprehensive proofs of the potential usefulness of fluorescence-based diagnostics during endoscopy of the oesophagus. In parallel we conducted other clinical studies of laryngeal lesions, also with convincing results.

C. Eker, S. Montan, E. Jaramillo, K. Koizumi, C. Rubio, S. Andersson-Engels, K. Svanberg, S. Svanberg, P. Slezak, Clinical spectral characterisation of colonic mucosal lesions using autofluorescence and delta aminolevulinic acid sensitisation, Gut, 44 (1999) 511-518. Cited by 95 Another endoscopic study, here of the gut, to investigate the potential usefulness of fluorescence-based detection techniques. Here the intention was to help find and classify diseased areas difficult to identify during conventional white-light endoscopy. This includes to distinguish premalignant and benign polyps and to find areas of carcinoma in situ (CIS).
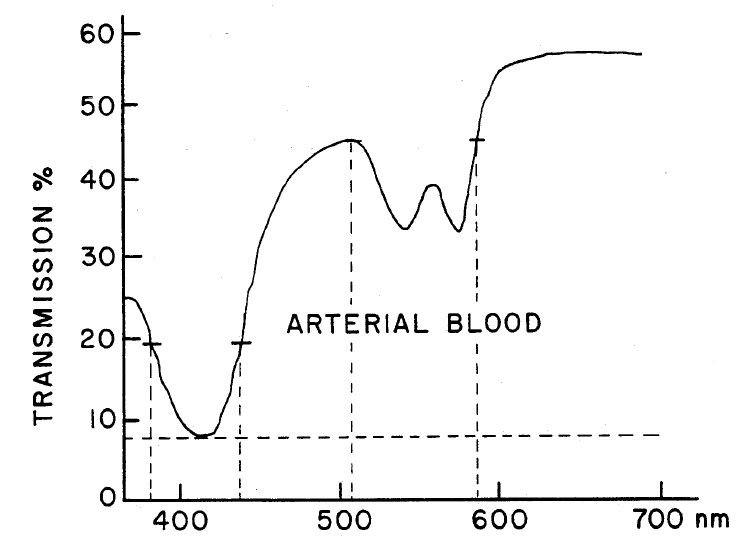
Diagnosis by means of fluorescent light emission from tissue, S Andersson-Engels, J Johansson, U Stenram, Katarina Svanberg, Sune Svanberg - US Patent 5,115,137, 1992. Cited by 165 This is an early patent to conduct multispectral fluorescence imaging with diagnostic potential based primarily on the intrinsic fluorescence properties by cleverly selected excitation or emission wavelengths.
A major barrier in optimising any light-based diagnostic signals or treatment dosimetry is the understanding of how light is scattered in biological tissue. Studying light interaction and propagation properties were of high importance, even in the early years.
In the early 1990s our pioneering work demonstrated the use of photon time-of-flight measurement to measure the optical properties in tissue. Our work led to the development of a photon time-of-flight spectroscopy (pTOFS) technique that has been applied widely since.
At the same time, I was also part of introducing durable tissue phantoms and our breakthrough research led to the MEDPHOT protocol to characterise systems to measure optical properties and I’m proud to say this protocol is still used today.
Recently, we introduced anthropomorphic phantoms to be able to conduct research aiming at developing a system to monitor the lung function in preterm infants.
This year I am excited to be co-founder of Tyndall spin-out known as BioPixS . This company aims to standardise the biophotonics instrument market by providing tissue phantoms and lasting standards for turbid media. Our vision is to translate cutting edge research into hi-tech innovative solutions to impact the BioPhotonics market.
Relevant research examples and selected publications include:
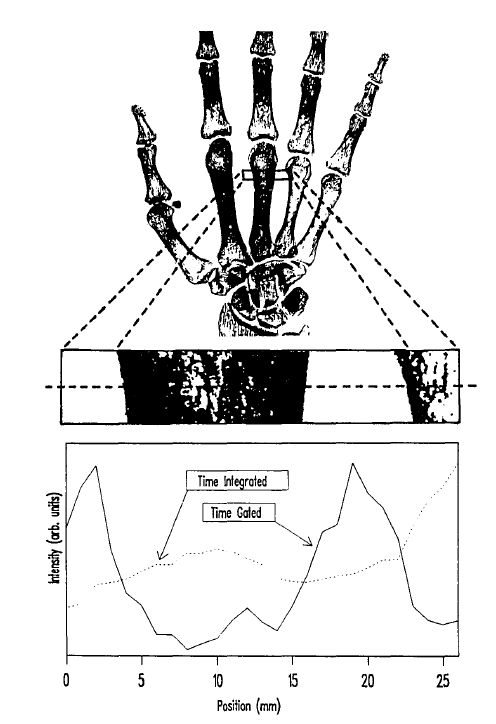
S. Andersson-Engels, R. Berg, S. Svanberg, O. Jarlman, Time-resolved Transillumination For Medical Diagnostics, Optics Letters, 15 (1990) 1179-1181. Cited by 348. This is a pioneering work on photon time-of-flight measurements in tissue. The study focuses on scanning applications to find areas of different optical properties. Implications for optical mammography are discussed.

S. Andersson-Engels, R. Berg, A. Persson, S. Svanberg, Multispectral Tissue Characterization with Time-resolved Detection of Diffusely Scattered White-light, Optics Letters, 18 (1993) 1697-1699. Cited by 92. By generating white high-intensity pulsed light by employing a high power pulsed laser and non-linear effects in water we here introduced pTOFS ex vivo and in vivo measurement.
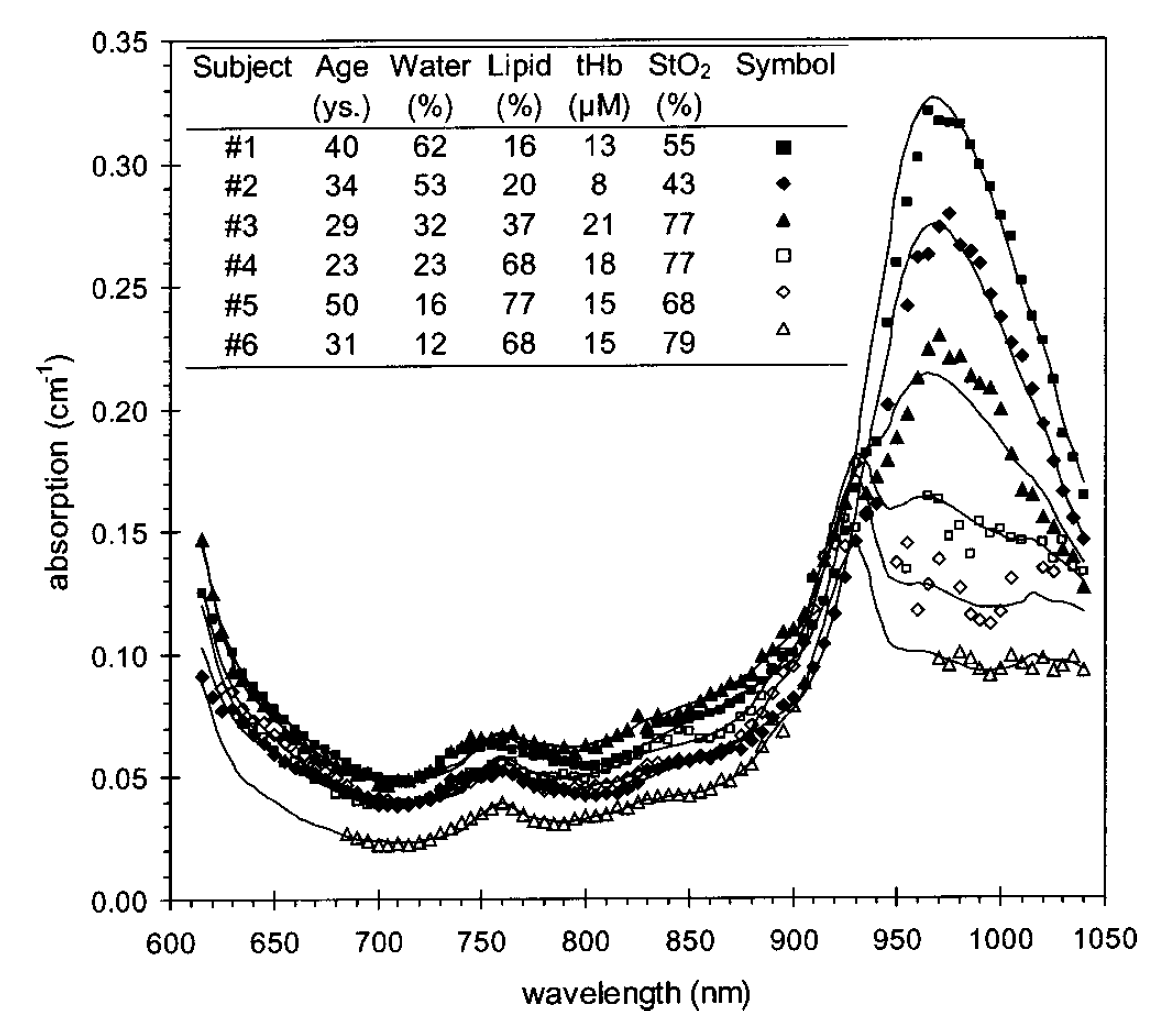
A. Pifferi, J. Swartling, E. Chikoidze, A. Torricelli, P. Taroni, A. Bassi, S. Andersson-Engels, R. Cubeddu, Spectroscopic time-resolved diffuse reflectance and transmittance measurements of the female breast at different interfiber distances, Journal of Biomedical Optics, 9 (2004) 1143-1151. Cited by 110. Once we developed the pTOFS technique to be better fitted for clinical measurements we conducted this in vivo breast tissue study. This work was conducted together with the group in Milan later becoming famous for further developing and employing this technique extensively. The adaption to clinical usability was accomplished by exchanging the high-power laser and water cuvette to generate the white light pulses, to a compact mode-locked fibre laser focused into a photonic crystal fibre.
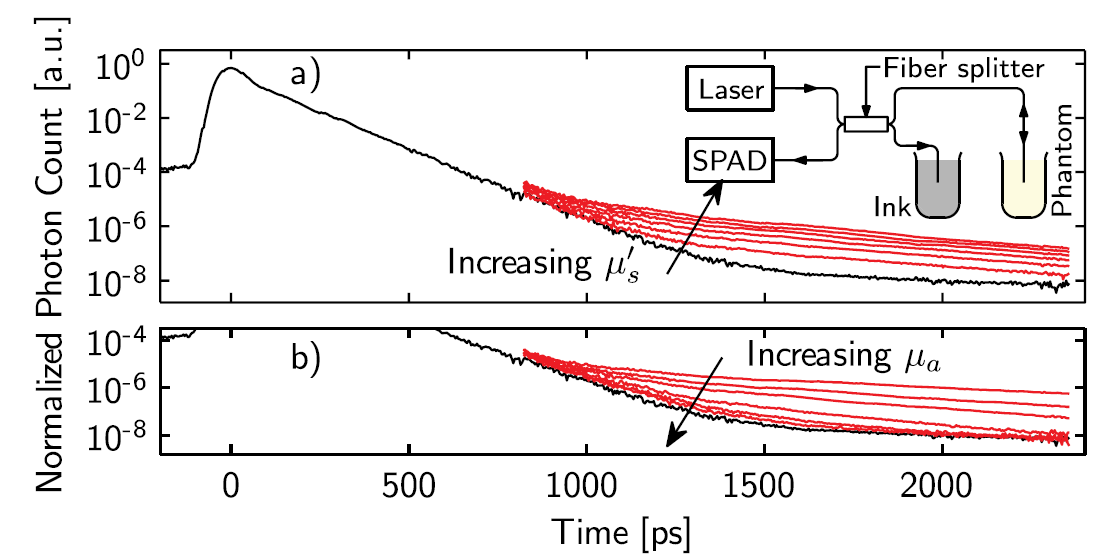
E. Alerstam, T. Svensson, S. Andersson-Engels, L. Spinelli, D. Contini, A. Dalla Mora, A. Tosi, F. Zappa, A. Pifferi, Single-fiber diffuse optical time-of-flight spectroscopy, Optics Letters, 37 (2012) 2877-2879. Cited by 39. This study was again conducted together with our close colleagues in Milan. We successfully demonstrated pTOFS to work with a single fibre. This is essential to be easily by employed for interstitial measurements, where a single fibre is very beneficial.
S.K.V. Sekar, P. Lanka, A. Farina, A. Dalla Mora, S. Andersson-Engels, P. Taroni, A. Pifferi, Broadband Time Domain Diffuse Optical Reflectance Spectroscopy: A Review of Systems, Methods, and Applications, Appl Sci-Basel, 9 (2019). This is a recent review of the field of photon time-of-flight spectroscopy written by people from Tyndall and Politecnico di Milano.
Knowing the optical properties of tissue allows us to calculate light propagation within tissue. I have been involved in developing and accelerating Monte Carlo modelling techniques for this purpose, and was leading research efforts which resulted in milestone papers in the field. These landmark discoveries accelerated the simulations several orders of magnitude in the problems we were solving. I have also been involved in research based on solving the wave equation of light transport in highly scattering media with e.g. Finite Difference Time Domain algorithms; as well as the diffusion equation to solve dosimetry problems for interstitial photodynamic therapy of prostate cancer.
Relevant research examples and selected publications include:

Nilsson, A., Alsholm, P., Karlsson, A., Andersson-Engels, S., "T-matrix computations of light scattering by red blood cells." Applied Optics 37(13): 2735-2748(1998). Cited by 145. The research described in this paper involved understanding of light transport in blood, of importance to develop optical spectroscopy techniques in whole blood.
Pifferi, P. Taroni, G. Valentini, S. Andersson-Engels, Real-time method for fitting time-resolved reflectance and transmittance measurements with a Monte Carlo model, Applied Optics, 37 (1998) 2774-2780. Cited by 104. In order to enable the use of slow Monte Carlo simulations for real-time fitting of measurements, we developed the method of white Monte Carlo simulations. This technique makes it possible to reduce the number of MC simulations for one for each absorption coefficient of the tissue, to one for all relevant values of the absorption coefficient.
J. Swartling, A. Pifferi, A.M.K. Enejder, S. Andersson-Engels, Accelerated Monte Carlo models to simulate fluorescence spectra from layered tissues, Journal of the Optical Society of America a-Optics Image Science and Vision, 20 714-727 (2003). Cited by 125. This paper introduces the concept of identic probabilities for light to be transported from A to B and from B to A. This identity was employed to accelerate fluorescence MC simulations by 3 orders of magnitude.
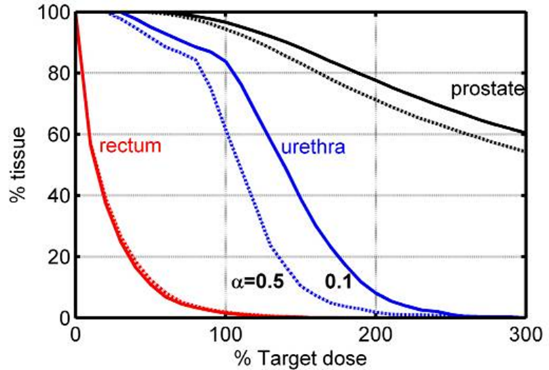
Johansson, J. Axelsson, S. Andersson-Engels, J. Swartling, Realtime light dosimetry software tools for interstitial photodynamic therapy of the human prostate, Medical Physics, 34 4309-4321 (2007). Cited by 93. Light transport in tissue is crucial in photodynamic therapy of malignant tumours. In this paper treatment “dose volume histograms” were calculated for the treatment of prostate cancer. Here a concept for intermittent measurements of the parameters important for the light dosimetry was employed to develop the treatment strategy that the spin-out company SpectrCure since is basing their interstitial PDT concept on.
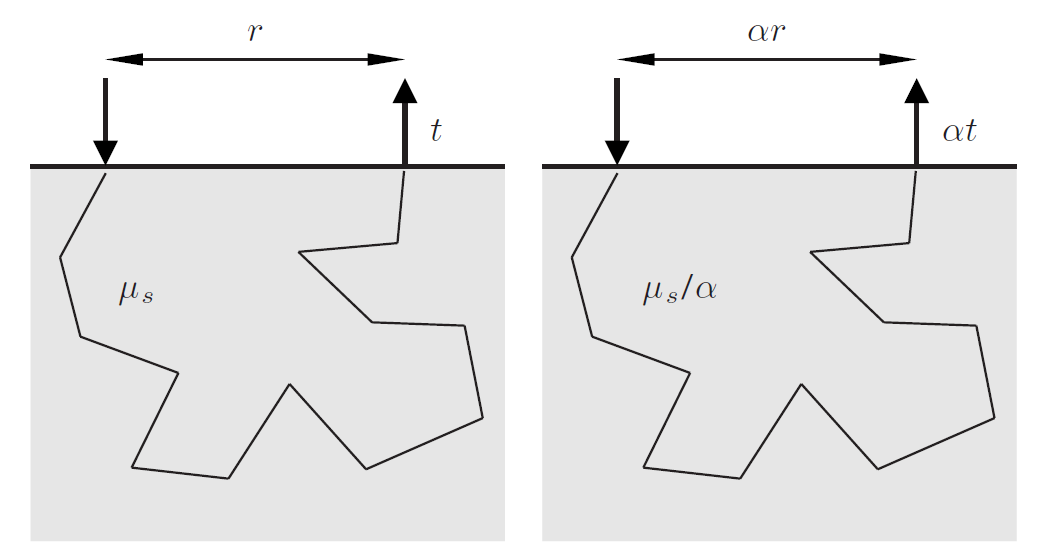
E. Alerstam, S. Andersson-Engels, T. Svensson, White Monte Carlo for time-resolved photon migration, Journal of Biomedical Optics, 13 (2008). Cited by 133. This is a paper where the white MC simulation concept was refined to become a useful tool for real-time data evaluation.
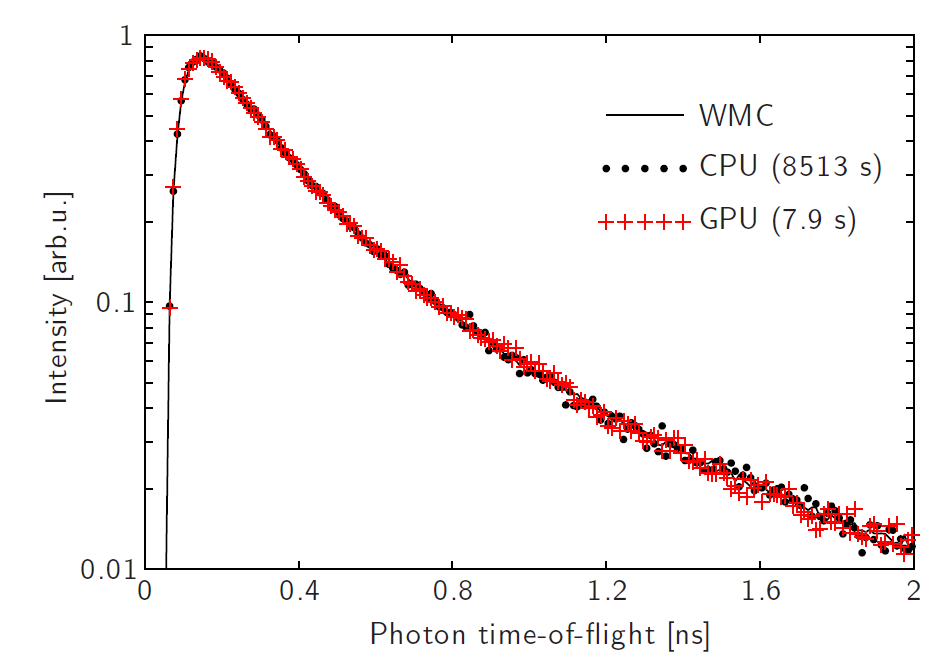
E. Alerstam, T. Svensson, S. Andersson-Engels, Parallel computing with graphics processing units for high-speed Monte Carlo simulation of photon migration, Journal of Biomedical Optics, 13 (2008). Cited by 422. Parallel computing has become a popular way to speed up computations. The paper describes the first MC simulations of light transport in biological tissue programmed for employing GPU processing. This can speed up simulations up to 3 orders of magnitude.
At Lund, we were the first to introduce photodynamic therapy of malignant tumours in Scandinavia 1987. Soon after, we were one of the first groups world-wide to use 5-amino levulinic acid instead of a porphyrin photosensitiser administered to the patient to catalyse the photodynamic reaction. We focussed on malignant skin cancer. Following this success, we soon started to consider interstitial light delivery to be able to treat thicker lesions. For this we needed to better understand the light penetration within the tumour tissue. In developing these techniques, I was one of three co-founders of the company SpectraCure .
Relevant research examples and selected publications include:
Svanberg, K., et al. (1994). "Photodynamic Therapy of Nonmelanoma Malignant-tumors of the Skin Using Topical Delta-amino Levulinic Acid Sensitization and Laser Irradiation." British Journal of Dermatology 130(6): 743-751. Cited by 492. This is one of the pioneering papers of photodynamic therapy of malignant skin tumours based on δ-amino levulinic acid (ALA). This has since become a treatment of choice for some tumours.
Wang, I., et al. (2001). "Photodynamic therapy vs. cryosurgery of basal cell carcinomas: results of a phase III clinical trial." British Journal of Dermatology 144(4): 832-840. Cited by 348. In this study we describe a clinical phase 3 study yielding very promising treatment results of ALA-PDT of malignant skin tumours.
Johansson, A., et al. (2007). "Realtime light dosimetry software tools for interstitial photodynamic therapy of the human prostate." Medical Physics 34(11): 4309-4321. Cited by 93. This paper describes the IDOSE algorithm employed in the interstitial PDT treatments now used in the SpectraCure technology.
Swartling, J., et al. (2010). "System for interstitial photodynamic therapy with online dosimetry: first clinical experiences of prostate cancer." Journal of Biomedical Optics 15(5). Cited by 78. The first clinical treatments of prostate cancer with interstitial PDT is described in this paper. This kind of treatment is now undergoing phase 2-3 clinical trials by SpectraCure.
Selected patent:

Therapy and diagnosis system and method with distributor for distribution of radiation, S Svanberg, S Andersson-Engels, K Svanberg, US Patent 7,037,325. Cited by 20. This is a first of a large number of patents owned by SpectraCure of how to conduct interstitial PDT with dosimetry and feedback from optical measurements and light transport calculations.
As one of the first research groups we were studying upconverting nanoparticles as a novel contrast agent for biomedical research in the mid-1990s. Early study together with our background in biomedical optics allowed us to publish papers that were critical in the development of a new field of research. This is a very active field of research today, and I am currently co-founder of a company called Lumito that uses such nanoparticles within the field of digital pathology www.lumito.se .
Relevant research examples and selected publications include:
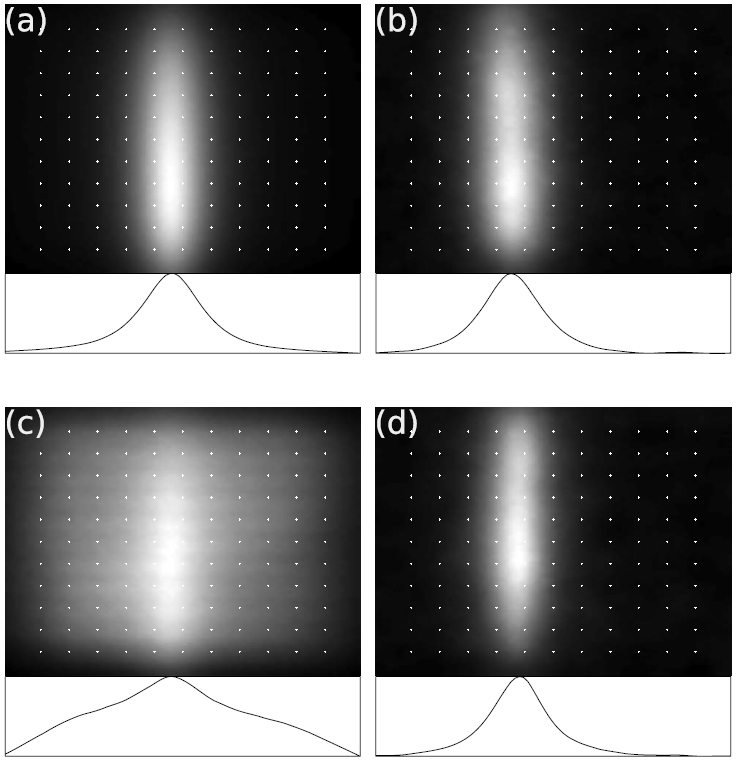
Xu, C. T., et al. (2008). "Autofluorescence insensitive imaging using upconverting nanocrystals in scattering media." Applied Physics Letters 93(17). Cited by 97. Here the main advantage of upconverting particles (UCNPs) as a contrast agent for imaging in biological tissue is proven – the background free high contrast signals. This paper was early illustrating the great potential of this unique method.
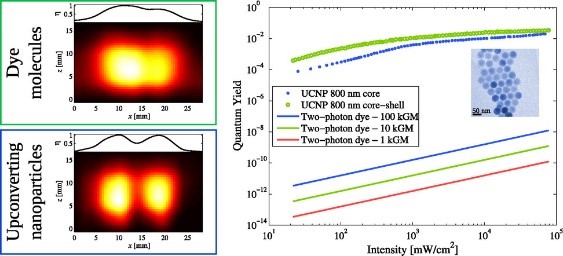
Xu, C. T., et al. (2012). "High-Resolution Fluorescence Diffuse Optical Tomography Developed with Nonlinear Upconverting Nanoparticles." ACS Nano 6(6): 4788-4795. Cited by 129. In this paper we employed the non-linear behaviour of UCNPs to illustrate the potential of improved spatial resolution in imaging.
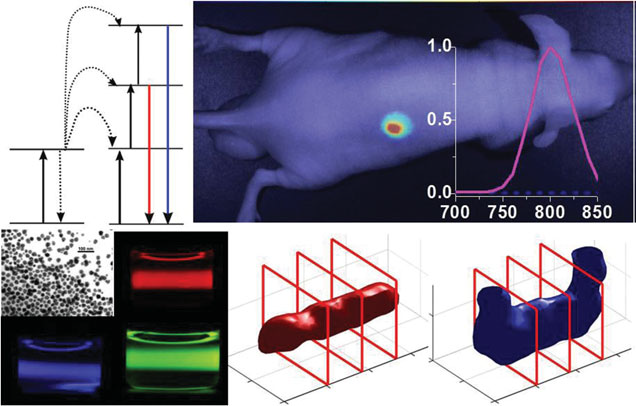
Xu, C. T., et al. (2013). "Upconverting nanoparticles for pre-clinical diffuse optical imaging, microscopy and sensing: Current trends and future challenges." Laser & Photonics Reviews 7(5): 663-697. Cited by 134. The early papers employing UCNPs for biological imaging were reviewed and potential of the field was outlined in this invited review.
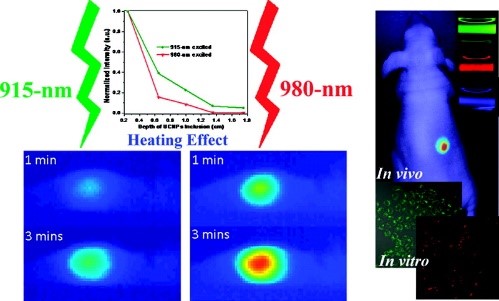
Zhan, Q. Q., et al. (2011). "Using 915 nm Laser Excited Tm3+/Er3+/Ho3+-Doped NaYbF4 Upconversion Nanoparticles for in Vitro and Deeper in Vivo Bioimaging without Overheating Irradiation." ACS Nano 5(5): 3744-3757. Cited by 451. UCNPs are favourable for biological tissue applications due to that the light of the wavelengths involved penetrate deep into tissue. Still the excitation wavelength at 975 nm is not fully optimal as water has a small absorption peak at this wavelength. In this paper we employed a shorter wavelength with lower absorption in the particles, but also by water. The results show lower side effect due to tissue heating during the imaging measurements.
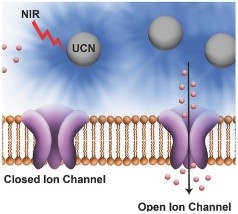
Bansal, A., et al. (2016). "Quasi-Continuous Wave Near-Infrared Excitation of Upconversion Nanoparticles for Optogenetic Manipulation of C-elegans." Small 12(13): 1732-1743. Cited by 56. In this paper we use UCNPs to locally generate the 470 nm light that is used for exciting the channel rhodopsin used for optogenetic research. This shows a great potential to use UCNPs to enable trans-dermal deep tissue optogenetics stimulation.
My research discoveries from the areas of fluorescence diagnostics, photon time-of-flight spectroscopy and tissue phantoms, modelling of light propagation in biological tissue and photodynamic therapy have instilled in me a deep-tech expertise that has enabled me to tackle significant, global, biomedical challenges and lead project teams that have produced impactful results along the way.
Early research in all of the above research fields has in effect produced real results, addressing biomedical challenges on a global scale, some of which are detailed below: I was early in utilising laser-induced fluorescence for in vivo studies, and many groups looked at our solutions to build mobile systems that could be brought to the clinic. I co-founded SpectraPhos for multispectral fluorescence imaging during endoscopy.
In photodynamic therapy, we introduced early - topically applied 5-ALA to treat skin cancer, a technique that is used in clinical practice today. The Lund group was sponsored by Johnson & Johnson to conduct a clinical randomised phase 3 trial to get the treatment approved.
I was also a co-founder of SpectraCure, a company that is conducting studies to get approval to treat prostate cancer with PDT, hopefully with less side-effects than current treatments.
I had a long collaboration with AstraZeneca to develop measurements techniques for the active substance in pharmaceutical drugs. We employed mainly time-of-flight measurements in these studies.
I was one of the authors of the first paper on the GASMAS technique. We are now employing this technique to develop a monitor tool for preterm infants. We are collaborating with GPX Medical, another spin-out of the Biomedical Optics activities at Lund University.
The successful research activity with upconverting nanoparticles made us believe that these particles could solve unmet needs in biophotonics. Therefore, we started the company Lumito, generating digital pathology images with superior contrast.
Since I was recruited to Cork with the help of strong engagement from IPIC, Tyndall and UCC, and financial support of SFI in form of a professorship award, I am collaborating with several companies to solve unmet needs. We are using our experience in biophotonics to find solutions to their challenges. In combination with the other expertise at IPIC we are in a very good position to find solutions that not only can solve their problems, but also be made compact and integrated in their existing tools and in the clinical work flow.
I have also engaged in making standards for biophotonics research and clinical use. In this context I was part of starting the company BioPixS Ltd. in 2020.
Teaching and training the next generation of motivated and talented individuals at all levels has always been privilege for me and was of course honoured to receive the prestigious “Lindbomska belöningen” teaching award from the Swedish Royal Academy of Sciences in Stockholm. I founded and co-organised the renowned international Biophotonics graduate summer school which has taken place annually since 2003. More than 500 PhD students from all continents have graduated from this school, grown professional networks as well as learned facts, methods and new skills. One attendee at the first school returned to the latest summer school as a lecturer. Due to my teaching engagements, I have been invited to lecture at approximately 20 other international summer schools as well as a travelling lecturer for student chapters of OSA and SPIE.
I have also been centrally engaged in developing and driving, as a valued teacher, the “International MSc program of Photonics” at Lund University and I am proud to have developed one of the first undergraduate courses worldwide in biomedical optics in 1997. The material of the course was openly shared via the internet, and has since been used in developing similar courses at many other universities worldwide (including several American Universities). I continue to engage as an advisor for MSc thesis projects, as indicated by being selected as a tutor by over 100 students for their half-to-one year projects.
Across my research team, I am a true advocate of a flat leadership philosophy. This means all team members are equally valued with no hierarchies in place, with the ambition to form an enthusiastic and creative atmosphere with many individual responsibilities. The advisor role for PhD students is thereby fundamentally changed, focusing on tutoring in the very early stages of a project to gradually allow increased initiative and responsibility to be undertaken towards the end of the project. The purpose behind this is to ensure PhDs are well equipped for a future career in academia or industry. I have supervised 34 PhD students to date, many of which have been successful in an academic career, both in physics and as clinical scientists. Other graduates have entered industry or continued with spin-off companies started during their studies.
Apart from my teaching skills, I have also been recognised for my entrepreneurial skills and have received several awards - including the SKAPA award, the most prestigious entrepreneurship award in Sweden. This was awarded for the idea behind one of the four spin-off companies which I cofounded: Spectraphos (founded in Lund 1989, later acquired by Canadian company), SpectraCure (founded in Lund 2003, board member 2003-2018, now valued at 150 M€), Lumito (founded in Lund 2011, ongoing board member since start, now valued at 28 M€), BioPixS Ltd (founded in Cork 2020, presently co-director, now valued at 3.5 M€). Among other notable successes, is an industrial collaboration project resulting in a filed patent and the 25-fold growth expansion of a small Med Tech company over the course of a year. I have also contributed to excellence by obtaining 20 patents awarded in 15 families.
Awards and honors
If you're interested in learning more about Stefan, visit his LinkedIn profile.